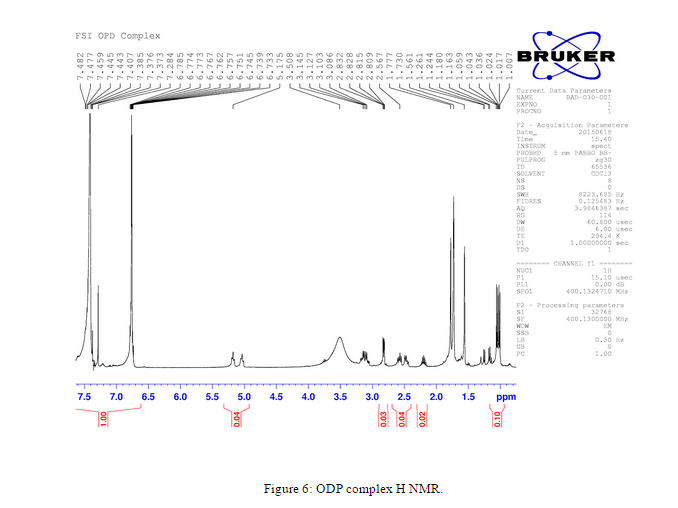
H NMR was taken after the o-phenaline diamine complex was isolated to verify that we had obtained the desired complex, Figure 6.
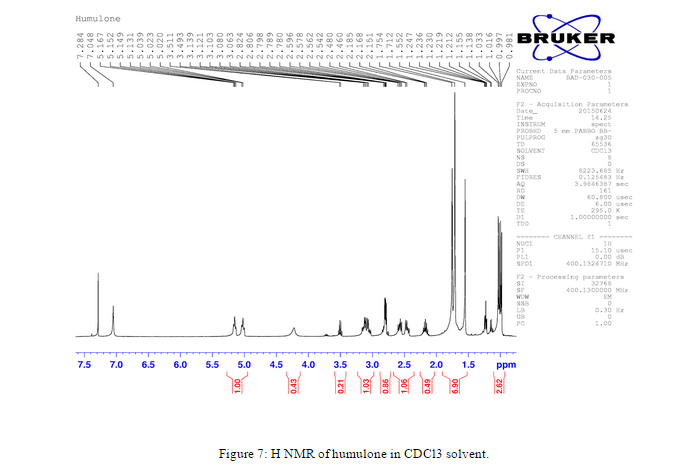
An H NMR was taken in deuterated chloroform after the isolation of humulone to verify that the complex was actually humulone, Figure 7.
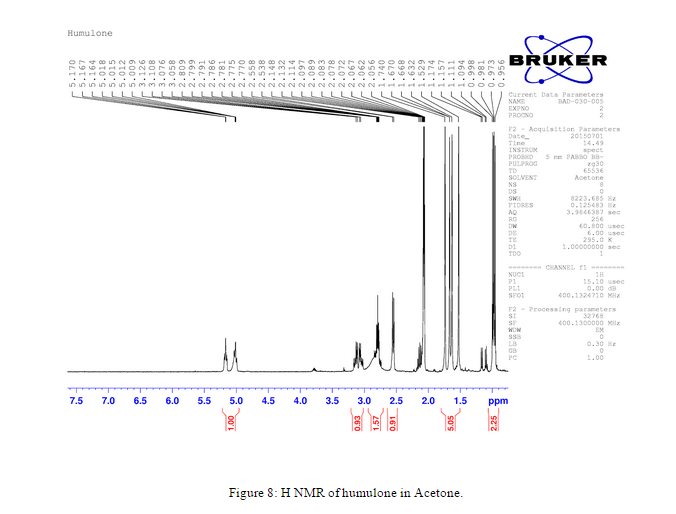
Another H NMR of humulone was taken of the humulone in deuterated acetone, Figure 8, instead of deuterated chloroform, Figure 7. We did this to compare the impurities of the H NMR peaks and the location of the solvent peaks in relation to the humulone peaks.
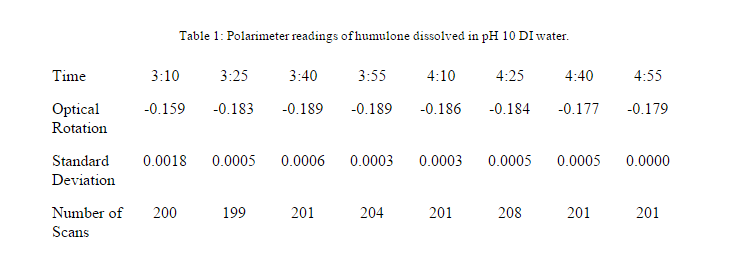
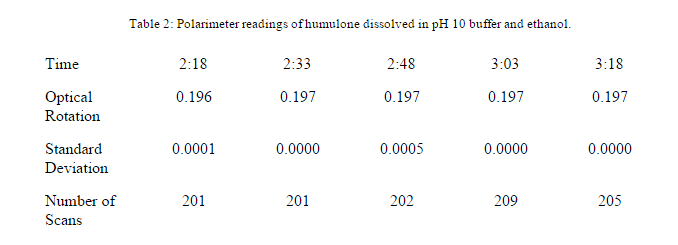
A polarimeter reading was taken for the humulone that was dissolved in DI water with a pH of 10.
A polarimeter reading was taken of the humulone dissolved in 8 mL of ethanol and 2 mL of pH 10 Ammonium Hydroxide Buffer.
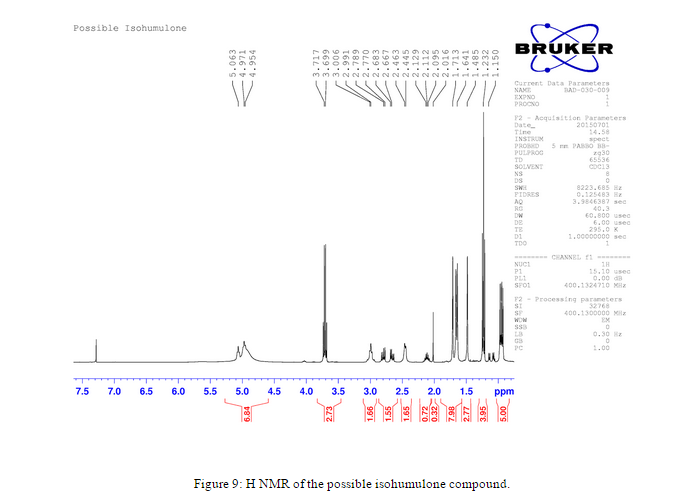
An H NMR was also taken of the resulting compound after the pH 10 Ammonium Hydroxide Buffer and ethanol solution was added, Figure 9. The resulting compound was believed to be a possible isohumulone.
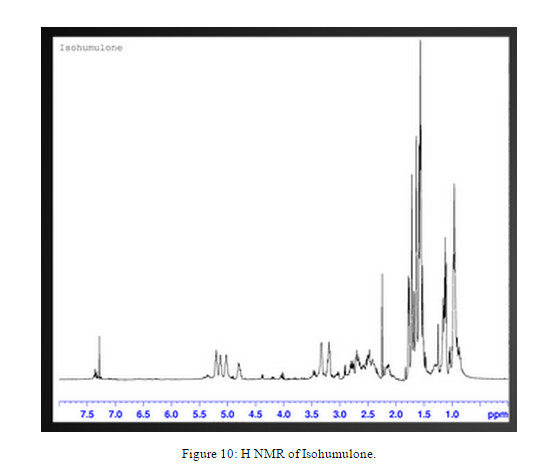
An H NMR was taken of the isolated isohumulone, Figure 10. Further purification of the compound is needed, but the differences between the isohumulone and possible isohumulone are still visible.