Beer has been brewed for thousands of years, but historically it has been more of an art than a science. Advancing technologies have begun to help people understand the chemical processes that occur during beer brewing. Modern beer got its start in the 14th century when hops, depicted below in Figure 1, began to be widely used for its preservative powers and flavor.
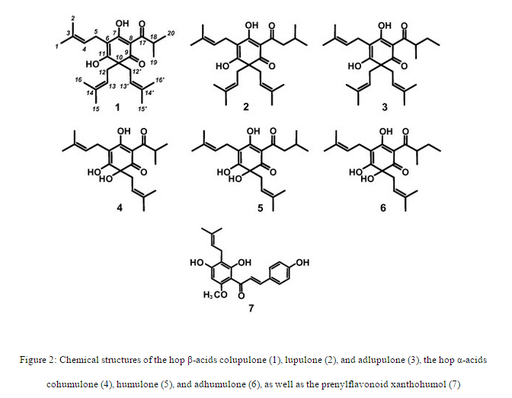
Two important compounds in hops are humulone and lupulone (Figure 2). These are often referred to as α-acids and β-acids respectively. Humulone was the main focus of our research. In the brewing process humulone is transformed into isohumulone when the wort is boiled. This transformation, known as an isomerisation, is when a compound changes into one of its structural isomers. A structural isomer has the same chemical composition, but it has a different structure, which gives it different chemical and physical properties. Isohumulone is significant because it provides the bitter flavor in beer and helps act as a preservative.
Isohumulone is a chiral molecule, so there are two types, cis- and trans- isohumulone. There are different types of chiral pairs. Cis- and trans- isohumulone are diastereomers. Unlike enantiomers, isohumulone has one arm that does not change direction in either form. Chiral molecules, like isohumulone, can rotate plane polarized light. The rotation of polarized light, light that is only on one plane, can be measured with a polarimeter, Figure 3. A polarimeter can be used to measure the change from humulone to isohumulone since the two rotate polarized light differently. It is our goal to identify the conditions in which isohumulone forms.